

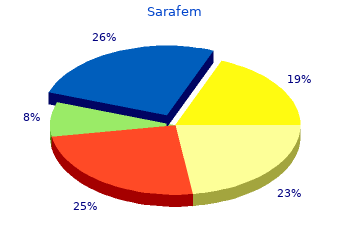
2018, Argosy University, Yasmin's review: "Sarafem 20 mg, 10 mg. Best Sarafem online no RX.".
Another study compared sitagliptin 100 mg/day with metformin 2000 mg/day (titrated up 38 over 5 weeks) trusted 10mg sarafem. Similar to the previous trial discussed cheap 20mg sarafem fast delivery, metformin resulted in greater HbA1c lowering (difference in LS mean change 0 discount sarafem 10mg otc. A 12 week dose-response study compared various doses of sitagliptin generic sarafem 20 mg on-line, all divided twice 30 daily, to glipizide titrated according to the study’s protocol. Slightly less HbA1c reduction was seen in the sitagliptin 100 mg/day group than the glipizide group. However, patients randomized to sitagliptin gained less weight than those in the glipizide group. Another study that stratified patients to sitagliptin 25 mg/day or 50 mg/ day based on their renal function, compared sitagliptin to placebo for the first 12 weeks and then glipizide 5-20 37 mg/day for the remaining 42 weeks. Patients in this study all had chronic renal insufficiency and were allowed to continue insulin therapy if on it prior to randomization. After 54 weeks, the placebo/glipizide group had slightly greater HbA1c reduction than sitagliptin. There was minimal change in HbA1c from 12 weeks to 54 weeks with sitagliptin. After 54 weeks, the sitagliptin group had greater weight loss than the placebo/glipizide group however after 12 weeks the placebo group had greater weight loss (Table 10) Results were not stratified by whether or not patients were taking insulin, however 7 patients in the sitagliptin group (11%) and 2 patients in the placebo group (8%) were on insulin at baseline. Efficacy outcomes of sitagliptin monotherapy compared with an active agent Change in HbA1c from baseline Change in weight from baseline at Author, year at (%) (kg) S25/50 PBO/Glip S25/50 PBO/Glip 54 weeks 54 weeks a 37 Chan, 2008 −0. Add-on therapy: Sitagliptin compared with active control (other oral hypoglycemic agent) added to metformin Four fair-quality trials (4 articles) compared the addition of sitagliptin with the addition of 34-36, 40 Pratley, 2010 #5847, 41 another oral hypoglycemic agent to ongoing metformin therapy. Pooled analysis was not conducted due to small number of studies with significant heterogeneity. One fair-quality trial compared the effects of adding either sitagliptin 100 mg/d or 34, 35 glipizide 5-20 mg/d in patients with inadequate glycemic control on metformin. Glycemic control was considered inadequate if the metformin dose was ≥ 1500 mg/d with baseline HbA1c 6. Over the initial 52 weeks the 2 study groups showed no significant differences in treatment effects for HbA1c (between-group difference 0. There was a statistically significant difference between treatment groups in the change in weight. Sitagliptin-treated subjects experienced slight weight loss) compared with a small weight gain seen in glipizide-treated subjects (between-group difference -2. Minimal changes were seen in HbA1c change from 52 week results and 104 week results, which is in contrast to a previously discussed trial and extensions 31-33 investigating sitagliptin as monotherapy. Prior to randomization patients had to have inadequate glycemic control (HbA1c 7% to 11%) and had to be taking metformin at stable doses ≥1500 mg/d for at least 10 weeks before entering a 2-week run-in period. In these patients, the addition of sitagliptin or rosiglitazone to metformin was significantly more effective than the addition of placebo to metformin at lowering HbA1c (P≤0. The placebo-corrected mean change from baseline was −0. Also, comparisons between sitagliptin and rosiglitazone were conducted and showed no statistically significant differences in lowering HbA1c (between-group difference: −0. Patients randomized to sitagliptin or placebo exhibited slight weight loss from baseline (sitagliptin, −0. Another trial compared the addition of colesevelan, rosiglitazone, or sitagliptin to 40 ongoing metformin. The trial found no statistically significant difference between the 40 rosiglitazone- and sitagliptin-treated subjects. An additional 26 week fair quality active-control trial compared liraglutide (1. All study participants were on metformin ≥ 1500 mg daily as background therapy. The study found a greater improvement in HbA1c with both doses of liraglutide compared to sitagliptin (change in HbA1c: liraglutide 1.
The difference in adverse events appeared to be related to differences in the rate of infusion reactions buy 20mg sarafem visa, although the 24-week duration of the study may not have been adequate to identify other differences 20mg sarafem amex. Children No evidence on the comparative safety of targeted immune modulators in children exists sarafem 10mg fast delivery. In the following paragraphs we summarize the scarce evidence that exists on the safety of targeted immune modulators in pediatric populations cheap 10 mg sarafem amex. Overall, various methodological issues limited the quality and applicability of this body of evidence. A major limitation was that the studies had small sample sizes and lacked power to detect rare but potentially serious adverse events. Furthermore, many of the studies used run-in periods, which seriously compromised the external validity of findings. During these run-in periods, with the active drug, only patients who responded, adhered to treatment, and had no intolerable adverse events were randomized to continuing active treatment or to placebo. Therefore, findings presented in the following paragraphs are subject to considerable uncertainty and should be interpreted accordingly. To provide a more realistic picture of the frequency of adverse events we focused on results from the open-label run-in phases that included a less selected population than the randomized phases. Five randomized controlled trials provided information on the general tolerability and safety of targeted immune modulators in children; four were in children with juvenile idiopathic 150 arthritis and one in children with plaque psoriasis. Drugs studied were abatacept, 152 153,253 154 adalimumab, etanercept, and infliximab. Generally, adverse event profiles in children were similar to those observed in adult populations. For example, in the adalimumab trial the 152 most common adverse events were infections and injection site reactions, which were also the most commonly reported adverse events in adult populations. During the open-label run-in phase of the adalimumab and methotrexate arm (n=85) the rate of any adverse event was 15. Targeted immune modulators 96 of 195 Final Update 3 Report Drug Effectiveness Review Project Injection site reactions (39% of children) and upper respiratory tract infections were the most commonly reported adverse events with etanercept during the run-in phase of a trial in 153 children with juvenile idiopathic arthritis. Nine children (15%) had to be hospitalized because 153,355 of serious adverse events during the 2-year extension phase. Fifty percent of the children received etanercept up to 4 years and the rate of serious adverse events in these children was 0. In the second trial of etanercept, in 211 children with plaque psoriasis, four serious adverse events occurred during the run-in phase treatment with etanercept including three 253 serious infections. Injection site reactions were reported to be mild and transient. In an uncontrolled trial of etanercept (n=60), 20% of children withdrew over a 12-month period 357 because of adverse events including severe infections, pancytopenia, and cutaneous vasculitis. In a case series based on data from a registry of children treated with etanercept in Austria and Germany (n=322) withdrawal rates because of adverse events were substantially lower than in 156 trials. Abatacept and infliximab are both administered intravenously and acute infusion reactions are a concern for both drugs. The rate of infusion reactions appeared to be greater in the infliximab study than in the abatacept study. Overall, 18% to 35% of children treated with 154 infliximab experienced acute infusion reactions. A case series of children (N=11) with 358 Crohn’s disease or ulcerative colitis reported infusion reactions in 8. By 150 comparison, only 4% of children on abatacept reported acute infusion reactions. A study of data from a registry of 202 children treated with infliximab as maintenance therapy for at least one year for Crohn’s disease reported one case of conversion of purified protein derivative skin test with a chest x-ray negative for tuberculosis, one case of varicella infection requiring hospitalization, one malignancy ultimately determined to be Hodgkin’s disease, and one death due to cardiac arrest (more than 6 months following discontinuation of 359 infliximab).